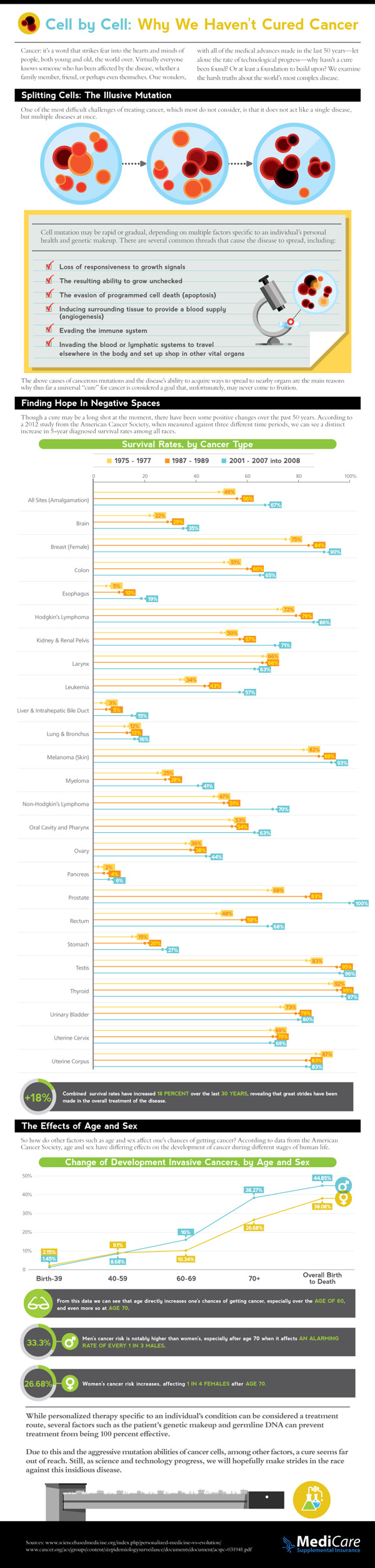
INDIANAPOLIS, /PRNewswire/ -- Eli Lilly and Company (NYSE: LLY) and Avid Radiopharmaceuticals, Inc., a wholly owned subsidiary of Lilly announced that Amyvid is available to imaging centers in markets surrounding 16 manufacturing sites located in Atlanta; Charlotte, N.C.; Chicago; Colton, Calif;Columbus, Ohio; Dallas; Fort Lauderdale, Fla.; Hartford, Conn.; Houston; Jacksonville, Fla.; North Wales, Pa.; Orlando, Fla.; Palo Alto, Calif.; Phoenix; Seattle; and St. Louis.
Because Amyvid loses over half of its radioactivity every two hours, it must be distributed directly to imaging centers from the specialized radiopharmacies where it is produced. Patients should speak to their health care provider to determine if Amyvid is an appropriate option for them and to determine if it is available in their area.
"In the roughly two months since the approval of Amyvid, we have been working closely with imaging centers to ensure they are prepared to provide Amyvid to their customers," said Daniel Skovronsky, M.D., Ph.D., president and CEO of Avid, and global brand development leader for Amyvid at Lilly. "We are committed to providing Amyvid to physicians, patients and their families in as many areas as possible as quickly as we can."
To coincide with the availability of Amyvid, Lilly is also announcing the launch of their reader training program. An online training program, developed by Lilly and Avid, is now available at AmyvidTraining.com. The companies also collaborated with the American College of Radiology to present a live, in-person training program, which will be launched on June 11, 2012, in Miami. These reader training programs were developed to provide training for radiologists and nuclear medicine physicians who will be responsible for reading Amyvid scans. Amyvid images should be interpreted only by readers who have successfully completed the Amyvid reader training. Errors may occur in the estimation of plaque density during image interpretation.
Amyvid is the first-and-only radioactive diagnostic agent approved by the U.S. Food and Drug Administration for PET imaging of the brain to estimate beta-amyloid neuritic plaque density in adult patients with cognitive impairment who are being evaluated for Alzheimer's Disease and other causes of cognitive decline.(1)
Amyvid works by binding to amyloid plaques, one of the necessary pathological features of Alzheimer's Disease,(2),(3),(4) and is detected using PET scan images of the brain.(1) A negative Amyvid scan indicates sparse to no amyloid plaques are currently present, which is inconsistent with a neuropathological diagnosis of Alzheimer's Disease and reduces the likelihood that a patient's cognitive impairment is due to Alzheimer's Disease.(2),(5) A positive Amyvid scan indicates moderate to frequent amyloid plaques are present; this amount of amyloid plaque is present in patients with Alzheimer's Disease, but may also be present in patients with other types of neurologic conditions and in older people with normal cognition.(1),(4),(6)
It's important to note that Amyvid is an adjunct to other diagnostic evaluations. A positive Amyvid scan does not establish a diagnosis of Alzheimer's Disease or other cognitive disorder. Additionally, the safety and effectiveness of Amyvid have not been established for predicting development of dementia or other neurologic condition, or monitoring responses to therapies.(1)
About Amyvid
Amyvid is a radioactive diagnostic agent, tagged with a radioisotope called fluorine-18. Once Amyvid is injected into a vein, it travels through the bloodstream and into the brain, binding to amyloid plaques. Amyvid produces a positron signal, which is detected by a PET scanner and used to create a brain image. A radiologist, who should have successfully completed Amyvid reader training, then interprets the image to evaluate for the presence or absence of significant amyloid plaques (i.e., moderate to frequent levels of neuritic plaques) in the brain. This information is reported back to the referring physician, who then determines the next steps in the evaluation and management of the patient.
Indications and Usage
Amyvid is indicated for Positron Emission Tomography (PET) imaging of the brain to estimate beta-amyloid neuritic plaque density in adult patients with cognitive impairment who are being evaluated for Alzheimer's Disease (AD) and other causes of cognitive decline.
A negative Amyvid scan indicates sparse to no neuritic plaques and is inconsistent with a neuropathological diagnosis of AD at the time of image acquisition; a negative scan result reduces the likelihood that a patient's cognitive impairment is due to AD. A positive Amyvid scan indicates moderate to frequent amyloid neuritic plaques; neuropathological examination has shown this amount of amyloid neuritic plaque is present in patients with AD, but may also be present in patients with other types of neurologic conditions as well as older people with normal cognition. Amyvid is an adjunct to other diagnostic evaluations.
Limitations of Use:
A positive Amyvid scan does not establish a diagnosis of AD or other cognitive disorder. Additionally, the safety and effectiveness of Amyvid have not been established for predicting development of dementia or other neurologic condition, or monitoring responses to therapies.(1)
Amyvid for intravenous use is supplied in 10 mL, 30 mL, or 50 mL multidose vials containing 500-1900 MBq/mL Florbetapir F 18.
Important Safety Information
Warnings and Precautions
Risk for Image Misinterpretation and Other Errors
Errors may occur in the Amyvid estimation of brain neuritic plaque density during image interpretation.
Image interpretation should be performed independently of the patient's clinical information. The use of clinical information in the interpretation of Amyvid images has not been evaluated and may lead to errors. Other errors may be due to extensive brain atrophy that limits the ability to distinguish gray and white matter on the Amyvid scan as well as motion artifacts that distort the image.
Amyvid scan results are indicative of the brain neuritic amyloid plaque content only at the time of image acquisition and a negative scan result does not preclude the development of brain amyloid in the future.
Radiation Risk
Amyvid, similar to other radiopharmaceuticals, contributes to a patient's overall long-term cumulative radiation exposure. Long-term cumulative radiation exposure is associated with an increased risk of cancer. Ensure safe handling to protect patients and health care workers from unintentional radiation exposure.
Most Common Adverse Reactions
The most common adverse reactions reported in clinical trials were headache (1.8 percent), musculoskeletal pain (0.8 percent), fatigue (0.6 percent), nausea (0.6 percent), anxiety (0.4 percent), back pain (0.4 percent), blood pressure increased (0.4 percent), claustrophobia (0.4 percent), feeling cold (0.4 percent), insomnia (0.4 percent), neck pain (0.4 percent).
Drug Interactions
Pharmacodynamic drug-drug interaction studies have not been performed in patients to establish the extent, if any, to which concomitant medications may alter Amyvid image results.
AM HCP ISI 06APR2012
About Eli Lilly and Company
Lilly, a leading innovation-driven corporation, is developing a growing portfolio of pharmaceutical products by applying the latest research from its own worldwide laboratories and from collaborations with eminent scientific organizations. Headquartered in Indianapolis, Ind., Lilly provides answers — through medicines and information — for some of the world's most urgent medical needs. Additional information about Lilly is available at www.lilly.com.
This press release contains certain forward-looking statements about Amyvid™, a radioactive diagnostic agent indicated for brain imaging of beta-amyloid plaques in patients with cognitive impairment who are being evaluated for Alzheimer's Disease and other causes of cognitive decline. This release reflects Lilly's current beliefs; however, as with any pharmaceutical product, there are substantial risks and uncertainties in the process of development and commercialization. There is no guarantee that future study results and patient experience will be consistent with study findings to date or that the product will prove to be commercially successful. For further discussion of these and other risks and uncertainties, see Lilly's filings with the United States Securities and Exchange Commission. Lilly undertakes no duty to update forward-looking statements.
P-LLY
(1) Amyvid Prescribing Information. April 6, 2012.
(2) Hyman BT, Phelps CH, Beach TG, et al. National Institute on Aging—Alzheimer's Association guidelines for the neuropathologic assessment of Alzheimer's disease. Alzheimers Dement. 2012;8:1—13.
(3) Mirra SS, Heyman A, McKeel D, et al; and participating CERAD neuropathologists. The Consortium to Establish a Registry for Alzheimer's Disease (CERAD): part II. standardization of the neuropathologic assessment of Alzheimer's disease. Neurology. 1991;41(4):479—486.
(4) Thies W, Bleiler L; Alzheimer's Association. Alzheimer's Association report: 2012 Alzheimer's disease facts and figures. Alzheimers Dement. 2012;8:131—168.
(5) McKhann GM, Knopman DS, Chertkow H, et al. The diagnosis of dementia due to Alzheimer's disease: recommendations from the National Institute on Aging-Alzheimer's Association workgroups on diagnostic guidelines for Alzheimer's Disease. Alzheimers Dement. 2011;7:263—269.
(6) Sperling RA, Aisen PS, Beckett LA, et al. Toward defining the preclinical stages of Alzheimer's disease: recommendations from the National Institute on Aging-Alzheimer's Association workgroups on diagnostic guidelines for Alzheimer's Disease. Alzheimers Dement. 2011;7(3):280—292.
©Lilly USA, LLC 2012. All rights reserved. AM77832 5/2012
Amyvid™ is a trademark of Eli Lilly and Company.
SOURCE Eli Lilly and Company